Furnaces and Instruments
Furnaces and Instruments
When Jacob Le Mort took over the chemical laboratory at the University of Leiden in 1690 on the death of Carel De Maets we are told that it contained the following items:
Van Spronsen, ‘The Beginning of Chemistry’ (1975).
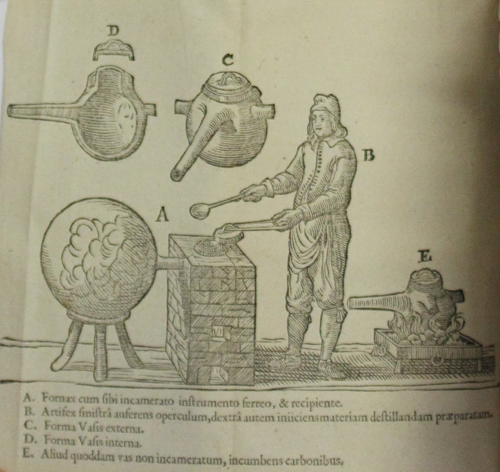
Johann Rudolf Glauber, Furni novi philosophici (Amsterdam, 1651), plate 2.
This illustration is from Worth’s copy of Johann Rudolph Glauber’s Furni novi philosophici, sive, Descriptio artis destillatoriae novae (Amsterdam, 1651), one of the most famous texts on furnaces written in the early modern period. He explains the illustration as follows: ‘A’ represents the furnace and the iron distilling wheel, to which a receiver is applied; ‘B’ is the distiller himself, who takes off the lid with his right hand in order to put in his prepared material with his left; ‘C’ represents the external form of the distilling vessel and ‘D’ is a cross section of its interior; ‘E’ represents another distilling vessel, which stands directly on a fire, rather than been attached to a furnace.
Glauber had taught both Carel de Maets and Jacob Le Mort and it was the latter who taught Edward Worth during his sojourn at the University of Leiden. Glauber divided his book into an analysis of five sections dealing with different types of furnaces and instruments: the first dealt with incombustible material; the second with combustible items; the third concentrated on distilling ‘burning spirits’; the fourth introduced a cheaper type of furnace and the fifth investigated other types of instruments. The popularity of Glauber’s text is unsurprising as it is very much a practical manual that would have been of use to any budding chymist. Glauber was interested above all in providing practical information about how furnaces were constructed, how they worked and what chymical operations they should be used for.
As his discourse on the second furnace illustrated above makes clear, he started first and foremost by describing how the furnace was built and how distilling took place, before moving on to specific preparations. The following is his account of the structure of his second furnace. For Glauber’s advice on how to distill using this furnace see the distillation webpage.
The Structure of the Second Furnace

Johann Rudolf Glauber, Furni novi philosophici (Amsterdam, 1651), plate 3.
By contrast, Glauber’s third furnace concentrated on copper and wood. His illustration for the third furnace presents us with a number of disparate images: in the top left of this image ‘A’ represents the ‘furnace with a Copper globe’; ‘B’ the Copper globe’ itself; ‘C’ the distilling vessel; ‘D’ the ‘refrigeratory with a worme’; ‘E’ the receiver’; and ‘F’ ‘stooles on which the vessels stand’. Beneath this structure we see two images: on the left ‘A Balneum with a cover having holes in it for the glasses, set upon a treefoot’. Beside it lies ‘a wooden vessel for the making of Beer’. To the right of these lie two types of bath: depicted above is ‘a Tub for a moyst Bath, which is to be warmed by the Copper Globe’; beneath this ‘a wooden Box for a dry Bath to provoke sweat with volatile spirits’.
Glauber explains how the third furnace is made in the following manner:
Glauber’s comments on this third sort of furnace are important for the insight they give us of chymistry as a social enterprise: he explicitly draws attention to the fact this wooden type of furnace was easily constructed by the poor and less costly than its counterparts, even if the new type of distillation process required a longer, slower fire than that necessary for other furnaces.

Johann Conrad Barckhausen, Pyrosophia… (Leiden, 1698), p. 68.
Johann Conrad Barchusen’s Pyrosophia contained a section on metallurgy and, as Hannaway (1967) observes, this was concerned with assay techniques. Barchusen was, in effect, introducing techniques to the university curriculum of Utrecht which were well known among miners. Hannaway lists the second section of this metallurgy course as follows:
- The separation of gold form silver by means of aqua fortis.
- As above by means of aqua regia.
- Purification of gold by cementation.
- Purification of gold per antimonium.
- Purification of gold with mercury.
- Purification of silver with lead.
As Hannaway argues, by doing this Barchusen was trying to broaden the boundaries of chemistry to extend beyond iatrochemistry.
Angelo Sala, Opera medico-chymica quae extant omnia (Frankfurt, 1647), 4R1r.
Angelo Sala (1576-1637), is perhaps best known as an early exponent of the corpuscular philosophy. As Clericuzio (2000) makes clear, Sala used corpuscular theory to explain chemical reactions. As a court physician to the dukes of Mechlenburg-Güstrow, Sala set up a laboratory at Güstrow. Worth’s copy of his Opera medico-chymica quae extant omnia (Frankfurt, 1647) contains a number of illustrations of the equipment he owned. Worth’s copy of Sala’s work was the first collection of Sala’s texts, many of which had gone through numerous printings in various languages from 1608 onwards.
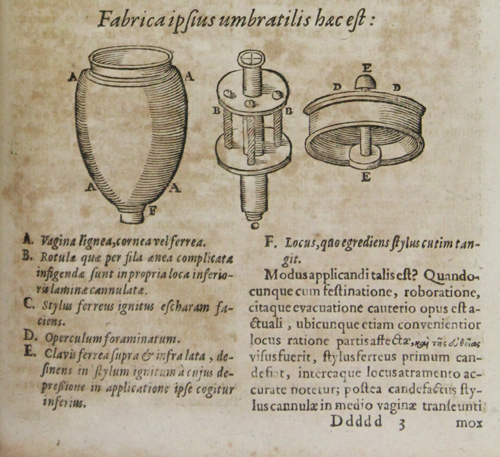
Sala instrument
Angelo Sala, Opera medico-chymica quae extant omnia (Frankfurt, 1647), 5D3r.
None of the experiments described here should be attempted.
Sources
Clericuzio, Antonio (2000), Elements, Principles and Corpuscles. A Study of Atomism and Chemistry in the Seventeenth Century (Kluwer).
Gelman, Zahkar E. (1994), ‘Angelo Sala, an iatrochemist of the late Renaissance’, Ambix, 41, (3), 142-160.
*Glauber, Johann Rudolph (1651), Furni novi philosophici (Amsterdam): the translation used here is the 1651 English translation: A Description of new Philosophical Furnaces… set forth in English, by J. F.D.M. (London, 1651).
Hannaway, Owen (1967), ‘Johann Conrad Barchusen (1666-1723) – contemporary and rival of Boerhaave’, Ambix 14, 96-111.
Holmes, Frederick L. and Levere, Trevor H. (eds.) (2002), Instruments and Experimentation in the History of Chemistry (MIT Press, Cambridge, Massachusetts).
Van Spronsen, J. W. (1975), ‘The Beginning of Chemistry’, in Lunsingh Scheurleer, Th. H. and Posthumus Meyjes, G. H. M. (ed.) Leiden University in the Seventeenth Century. An Exchange of Learning (Leiden), pp 329-343.


